The delicate touch of a princess discerning a minuscule pea beneath a mountain of mattresses, a cornerstone of fairy tale lore, finds a surprising echo in the realm of cellular biology. For decades, scientists have marveled at the ability of certain cells, particularly those exhibiting aberrant behavior like cancer cells, to perceive their environment far beyond their immediate physical contact. Now, groundbreaking research from Washington University in St. Louis is illuminating a previously underappreciated phenomenon: ordinary cells, when acting in concert, can achieve a comparable, and even amplified, long-range environmental sensing capability. This discovery, detailed in the esteemed journal PNAS, not only deepens our understanding of fundamental cellular mechanics but also holds significant promise for unraveling the intricate processes of cancer metastasis and developing novel therapeutic strategies.
The Phenomenon of Depth Mechano-Sensing
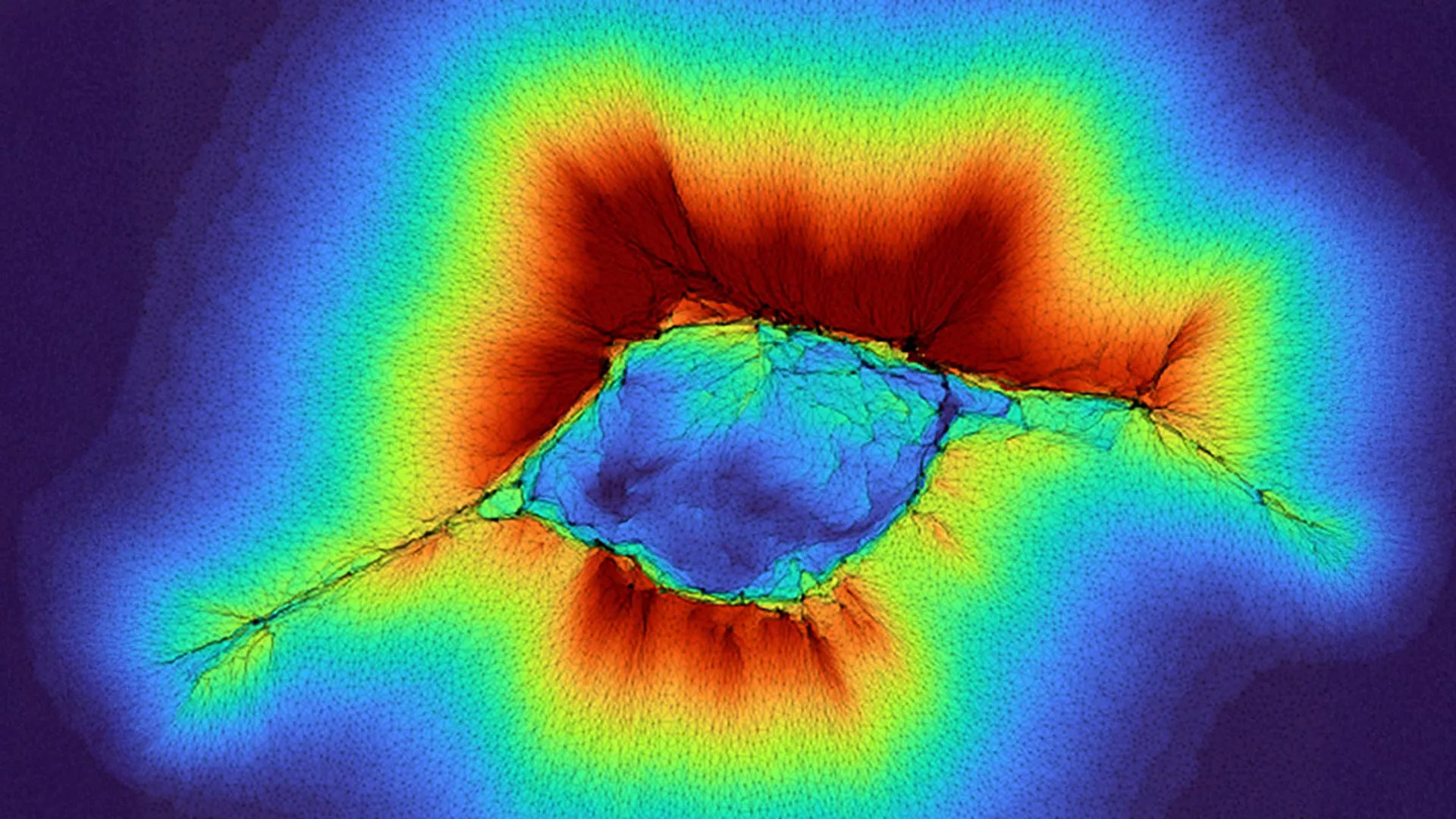
At the heart of this discovery lies the concept of "depth mechano-sensing," a term coined to describe a cell’s capacity to detect physical cues and properties situated beyond the surface to which it is adhered. Dr. Amit Pathak, a professor of mechanical engineering and materials science at Washington University’s McKelvey School of Engineering, has been a leading figure in investigating how cells interact with the physical attributes of their surroundings. His previous work had already established that cells with a pronounced "front-rear polarity," a characteristic often associated with migrating cells, possess a heightened ability to sense their environment. These abnormal cells, Pathak and his team had previously demonstrated, could detect physical features extending up to 10 microns away from their attachment point.
This remarkable sensory acuity in individual, migrating cells is partly attributed to their interaction with the extracellular matrix (ECM), a complex network of fibrous proteins, primarily collagen, that provides structural support and biochemical cues to cells. As a cell moves, it exerts forces on these surrounding collagen fibers, pulling and deforming them. This deformation effectively extends the cell’s "reach," allowing it to probe and "feel" the composition and stiffness of subsequent layers of the ECM. The stiffness of these layers – whether it be the dense, unyielding structure of a tumor, the pliant nature of soft tissue, or the rigidity of bone – provides critical directional information, guiding the cell’s migratory path. This ability to discern mechanical properties from a distance is crucial for processes like tissue development, wound healing, and, unfortunately, the invasive spread of cancer.
Collective Forces Amplify Sensing Range
The recent findings by Pathak’s laboratory represent a significant leap forward by demonstrating that this depth mechano-sensing is not solely the domain of specialized or aberrant cells. Their research unequivocally shows that ordinary epithelial cells, the fundamental building blocks of many of the body’s surfaces, can collectively achieve an astonishingly greater sensing range. When these cells cooperate and act as a unified front, they generate amplified forces that enable them to probe through the fibrous collagen network and detect environmental features as far as 100 microns away. This represents a tenfold increase in the sensing range compared to individual migrating cells.
"Because it’s a collective of cells, they are generating higher forces," Dr. Pathak explained, highlighting the crucial role of multicellular cooperation in this enhanced sensing capability. The research, co-authored by PhD student Hongsheng Yu, employed sophisticated computational modeling to elucidate the underlying mechanisms. These models suggest that the process unfolds in distinct stages as cells begin to cluster and initiate collective migration. During these critical phases, the environmental information gathered through this amplified sensing mechanism directly influences the subsequent movements and spreading patterns of the cell group.
Implications for Understanding and Combating Cancer Metastasis
The implications of this discovery for cancer research are profound and far-reaching. Cancer cells, with their inherent migratory tendencies, appear to exploit this enhanced collective sensing ability to their advantage. Their capacity to "see" ahead, to detect the mechanical landscape of the surrounding tissue, allows them to navigate away from the primary tumor environment and infiltrate adjacent tissues. This advanced awareness of their surroundings enables them to move more effectively, even through softer tissue environments, and crucially, helps them evade the body’s natural detection mechanisms.
The ability of cancer cells to metastasize – to spread from their primary site to distant parts of the body – is the primary driver of cancer-related mortality. Understanding the intricate mechanisms that facilitate this spread is therefore paramount in the fight against cancer. This new research offers a crucial piece of that puzzle by revealing how even groups of seemingly ordinary cells, when acting collectively, can exhibit a sophisticated ability to navigate complex biological terrain.
Researchers are now keen to delve deeper into the precise control mechanisms governing this extended sensing range. The next critical step involves identifying specific molecular regulators that dictate how far cells can perceive their environment. The discovery of these regulators could pave the way for the development of entirely new therapeutic avenues. If scientists can devise strategies to disrupt a cancer cell’s ability to "feel" its path forward, they may be able to effectively inhibit its migratory capabilities, thereby limiting the spread of the disease and improving patient outcomes.
A Timeline of Discovery and Future Directions
The research leading to these findings represents a culmination of years of dedicated study into cellular mechanics and behavior. While the exact start date of the specific project leading to the PNAS publication is not publicly detailed, the foundational work on depth mechano-sensing in migrating cells by Dr. Pathak’s lab can be traced back to earlier investigations. These prior studies, which established the 10-micron sensing range in individual abnormal cells, likely occurred within the past five to seven years, given the typical pace of scientific advancement and publication cycles in this field.
The transition to exploring collective cell behavior and its impact on sensing range would have followed logically, building upon the established principles. The computational modeling component, crucial for understanding the staged process of collective sensing, suggests a significant investment in computational resources and expertise, likely over the past two to three years. The PNAS publication itself marks a significant milestone, representing peer-reviewed validation of these complex findings.
Looking ahead, the research trajectory is clear. The immediate focus will be on pinpointing the specific molecular pathways and protein interactions that mediate this collective depth mechano-sensing. This could involve a range of experimental techniques, including advanced microscopy, genetic manipulation of cell lines, and biomechanical assays. Furthermore, researchers will likely seek to validate these findings in more complex three-dimensional cellular models that more closely mimic the in vivo environment.
Broader Impact and Potential Applications
Beyond cancer, the implications of enhanced collective cell sensing could extend to other areas of biological research and regenerative medicine. Understanding how groups of cells coordinate their movements and environmental perception is fundamental to tissue engineering, where the goal is to create functional tissues and organs from cellular components. If researchers can harness or even mimic this collective sensing ability, it could lead to more efficient and precise methods for guiding cell behavior in engineered tissues.
For instance, in regenerative medicine, promoting the coordinated migration and integration of cells into damaged tissues is a critical challenge. A deeper understanding of how collective sensing influences cell-cell interactions and tissue integration could inform strategies for accelerating wound healing or repairing damaged organs.
The funding for this significant research, provided by the National Institutes of Health (NIH) under grant R35GM128764 and the National Science Foundation’s Civil, Mechanical and Manufacturing Innovation program (grant 2209684), underscores the perceived importance of this line of inquiry by major scientific funding bodies. These grants represent a substantial investment in fundamental biological research with clear translational potential.
Expert Reactions and Future Outlook
While direct statements from external parties are not included in the original report, the scientific community’s reaction to such a fundamental shift in understanding is typically one of cautious optimism and intellectual engagement. Leading researchers in cell biology, biomechanics, and cancer research would likely view these findings as a pivotal advancement. Discussions at upcoming scientific conferences in fields such as cell mechanics, cancer biology, and tissue engineering will undoubtedly feature this work prominently.
The implications for future drug development are particularly noteworthy. Current cancer therapies often target specific molecular pathways within cancer cells. However, this research suggests that a more promising approach might involve targeting the physical interactions and collective behaviors that enable cancer cells to spread. Developing drugs that interfere with the collective mechanical forces generated by cancer cell clusters, or that block the sensing mechanisms that guide their migration, could represent a novel and potentially more effective strategy to combat metastatic disease.
In conclusion, the research from Washington University in St. Louis has unveiled a remarkable capacity within ordinary cells to sense their environment at distances previously thought to be exclusive to specialized or pathological cell types. By demonstrating that collective action amplifies this sensing range exponentially, scientists have opened new avenues for understanding fundamental biological processes and have provided a critical new perspective in the ongoing battle against cancer. The "princess-like sensitivity" of cells, it turns out, is not a solitary trait but a powerful emergent property of cellular cooperation.