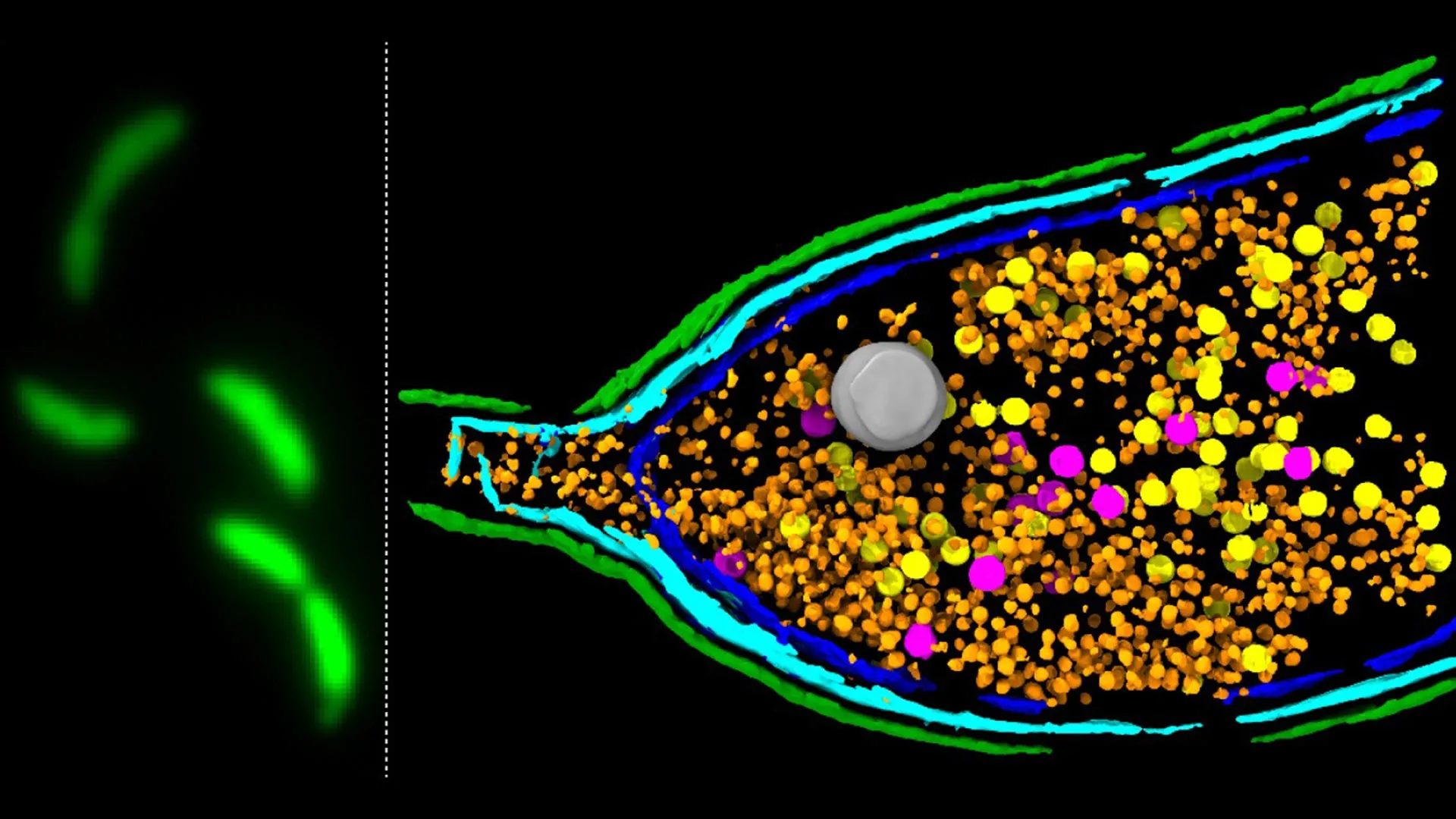
Scientists have unveiled groundbreaking insights into the clandestine world of bacterial gene sharing, revealing critical mechanisms that underpin the rapid dissemination of antimicrobial resistance (AMR), a formidable and escalating global health crisis. Researchers at the esteemed John Innes Centre, in collaboration with leading institutions, have meticulously dissected the function of unusual particles known as gene transfer agents (GTAs), shedding new light on how these microscopic couriers facilitate the exchange of vital genetic material between bacteria. This pivotal discovery, published in the prestigious journal Nature Microbiology, not only deepens our understanding of fundamental bacterial biology but also offers crucial clues in the ongoing battle against drug-resistant pathogens.
The Mystery of Gene Transfer Agents: Unlocking Bacterial Communication
For years, the scientific community has recognized that bacteria possess sophisticated methods for exchanging genetic information, a process that dramatically accelerates their adaptation and survival. Among these mechanisms, horizontal gene transfer (HGT) stands out, allowing bacteria to acquire new traits from unrelated individuals, a stark contrast to the vertical gene transfer observed in reproduction. Gene transfer agents (GTAs) have emerged as key players in this phenomenon. These enigmatic particles, structurally reminiscent of bacteriophages – viruses that infect bacteria – are not, however, parasitic invaders. Instead, they represent a remarkable evolutionary adaptation, derived from ancient viral genomes that bacteria have domesticated and repurposed for their own benefit.
GTAs operate as highly efficient biological delivery systems. They are meticulously engineered by the host bacterium to encapsulate fragments of its own DNA. These DNA parcels are then packaged within the GTA particle, which subsequently buds off from the cell or is released upon cell lysis. Once outside, these GTA particles can readily infect neighboring bacterial cells, delivering their genetic cargo. This transfer can introduce a wide array of genes, including those that confer resistance to antibiotics, rendering previously effective treatments obsolete. The speed and efficiency of this process underscore the urgency of understanding its underlying molecular machinery.
The Crucial Step: Host Cell Lysis and GTA Release
A critical bottleneck in the GTA life cycle, and a long-standing puzzle for researchers, has been the precise mechanism by which these particles are released from the host cell. This process, known as host cell lysis, involves the controlled rupture of the bacterial membrane, creating an exit route for the newly formed GTA progeny. Without efficient lysis, the dissemination of genetic material via GTAs would be severely hampered, limiting the bacteria’s ability to spread advantageous traits.
The research team at the John Innes Centre, employing a sophisticated deep sequencing-based screening methodology, has successfully pinpointed the genetic components responsible for orchestrating this vital step. By systematically analyzing the genome of the model bacterium Caulobacter crescentus, known for its well-characterized GTA system, they were able to identify a specific gene cluster with profound implications for GTA release.
The LypABC System: A Master Regulator of Cell Lysis
The investigation led to the identification of a pivotal three-gene system, dubbed LypABC. This genetic locus encodes a set of bacterial proteins that, when expressed, are directly involved in triggering the lysis of the host cell. The researchers conducted rigorous experiments to elucidate the role of LypABC. When the lypABC genes were experimentally deleted from the bacterial genome, the cells were observed to be incapable of lysing and consequently failed to release GTA particles. This finding unequivocally demonstrated that the LypABC system is essential for GTA production and egress.
Conversely, when the researchers artificially amplified the expression of the lypABC genes, a significantly higher proportion of bacterial cells underwent lysis. This dose-dependent response further solidified the conclusion that LypABC acts as a central control hub, dictating the timing and extent of cell rupture for GTA release. The precise molecular choreography orchestrated by LypABC ensures that GTA production is coupled with the necessary cellular breakdown for their effective dispersal.
An Unexpected Repurposing: Immune System Components at Work
Perhaps one of the most surprising and illuminating discoveries of this research lies in the origin and nature of the LypABC system. Astonishingly, the protein components encoded by lypABC bear a striking resemblance to a well-established bacterial defense mechanism: an anti-phage immune system. Bacterial immune systems, often involving protein complexes like CARD-NLRs (in some systems), are typically deployed to detect and neutralize invading viruses. However, in the context of GTA production, these same protein domains appear to have been evolutionarily repurposed.
This repurposing suggests a fascinating evolutionary narrative. Ancient viral elements, likely once pathogenic, were integrated into the bacterial genome. Over time, bacteria evolved mechanisms to control these elements, eventually harnessing them for their own reproductive and adaptive advantage. The LypABC system, therefore, represents a sophisticated example of how bacteria can ingeniously re-engineer existing biological machinery, transforming a defense system into a tool for genetic dissemination. This collaborative work, involving expertise from the University of York and the Rowland Institute at Harvard, underscores the remarkable plasticity and adaptability of bacterial biological systems.
The Imperative of Tight Regulation: A Double-Edged Sword
The researchers also identified a crucial regulatory protein that plays a vital role in maintaining stringent control over GTA activity. This regulatory element is indispensable because the LypABC system, while essential for gene transfer, can be inherently toxic to bacterial cells if its activation is not precisely managed. Uncontrolled or premature lysis would not only be detrimental to the individual bacterium but could also lead to the premature release of immature or non-viable GTA particles, compromising the efficiency of gene transfer.
This discovery highlights the delicate evolutionary balancing act that bacteria perform. They must possess the capacity to efficiently share genes for rapid adaptation, but this capability must be tightly regulated to ensure the survival of the host cell and the integrity of the genetic material being transferred. The regulatory protein acts as a critical safeguard, ensuring that GTA production and release occur at the optimal time and under appropriate conditions.
Broader Implications: A Deeper Understanding of AMR Spread
The implications of this research extend far beyond a fundamental understanding of bacterial genetics. The rapid spread of antimicrobial resistance (AMR) is a pressing global health emergency, threatening to undermine decades of progress in modern medicine. Bacteria acquire resistance genes through various means, including HGT. By elucidating the intricate mechanisms of GTA-mediated gene transfer, scientists gain invaluable insights into one of the primary drivers of AMR dissemination.
"What’s particularly interesting is that LypABC looks like an immune system, yet bacteria are using it to release GTA particles," stated Dr. Emma Banks, the study’s first author and a Royal Commission for the Exhibition of 1851 Research Fellow. "It suggests that immune systems can be repurposed to help bacteria share DNA with each other — a process that can contribute to the spread of antibiotic resistance." This quote emphasizes the surprising evolutionary pathways bacteria employ and directly links these fundamental discoveries to the AMR crisis.
The findings provide a mechanistic explanation for how resistance genes can move with surprising speed through bacterial populations. Understanding these pathways is crucial for developing novel strategies to combat AMR. This could involve designing compounds that inhibit GTA production or release, thereby slowing down the spread of resistance genes. It could also involve leveraging our knowledge of these systems to engineer bacteria that are less prone to acquiring resistance.
The Road Ahead: Unraveling Activation Pathways
The research has successfully identified the core machinery responsible for GTA release, but crucial questions remain. The next phase of this investigation will focus on unraveling the intricate details of how the LypABC system is activated and precisely how it orchestrates the rupture of bacterial cells to ensure the efficient release of GTA particles. Understanding the signaling cascades and environmental cues that trigger LypABC activation will be critical for a complete picture of this gene transfer process.
This ongoing work, building upon the foundational discoveries published in Nature Microbiology, promises to yield further insights into the remarkable adaptability of bacteria and their ability to evolve in response to selective pressures, including the widespread use of antibiotics. By demystifying these sophisticated genetic exchange mechanisms, scientists are better equipped to confront the escalating threat of antimicrobial resistance and safeguard public health for generations to come. The study, "A bacterial CARD-NLR-like immune system controls the release of gene transfer agents," represents a significant leap forward in our comprehension of these vital processes.