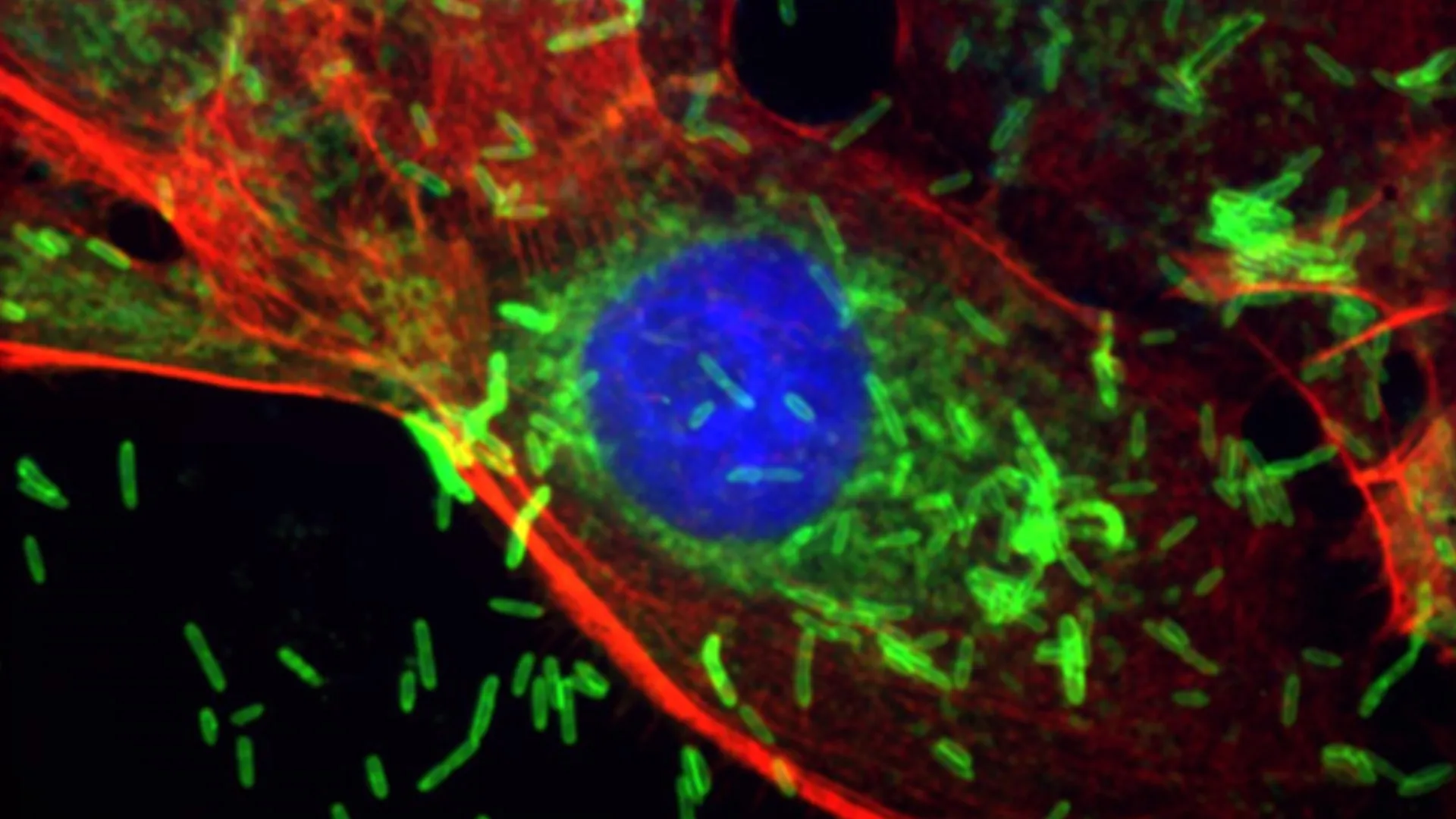
Scientists at the Johns Hopkins Kimmel Cancer Center and the Bloomberg~Kimmel Institute for Cancer Immunotherapy have identified a surprising and significant link between oral health and breast cancer. Their groundbreaking research reveals that Fusobacterium nucleatum, a bacterium most commonly associated with periodontal disease, may play a direct role in initiating breast cancer, accelerating tumor growth, and promoting its metastasis. This occurs through mechanisms that damage DNA and fundamentally alter the behavior of cancer cells, according to findings published in the esteemed journal Cell Communication and Signaling.
The study, building upon a growing body of epidemiological evidence that has observed correlations between periodontal disease and an increased risk of breast cancer, sought to unravel the specific biological pathways involved. Previous research, analyzing data from thousands of patients, consistently pointed to a connection, prompting the Johns Hopkins team to delve deeper into the molecular mechanisms at play. The implications of this research extend beyond understanding cancer development, potentially paving the way for novel diagnostic and therapeutic strategies rooted in oral hygiene.
Unveiling the Microbial Culprit: Fusobacterium nucleatum
Fusobacterium nucleatum is a ubiquitous bacterium found in the human oral cavity, typically residing in the gums. It is a key player in the development of periodontitis, a severe form of gum disease characterized by inflammation of the gums and damage to the bone and tissues that support the teeth. While its role in oral health is well-documented, this new research highlights its sinister potential beyond the mouth.
The Johns Hopkins team discovered that F. nucleatum is not confined to the oral cavity. It can, under certain circumstances, enter the bloodstream and migrate to distant sites, including breast tissue. Once established in breast tissue, the bacterium appears to act as a potent instigator of the carcinogenic process.
"The key takeaway is that this oral microbe can reside in breast tissue and that there is a connection between this pathogen and breast cancer," stated Dr. Ajit Sharma, a senior author on the study and a researcher at the Bloomberg~Kimmel Institute for Cancer Immunotherapy. "We were inspired by many small studies that looked at thousands of patients and connected periodontal disease to breast cancer. We wanted to dig deeper and see if we could uncover the underlying connections."
This sentiment was echoed by Dr. Sheetal Parida, Ph.D., the first author of the study and a research associate working with Dr. Sharma. "Our goal was to move beyond correlation and establish a causal link, understanding precisely how this bacterium influences the complex journey of breast cancer development," Dr. Parida remarked.
Mechanisms of Harm: DNA Damage and Tumor Acceleration
The research employed a combination of in vitro experiments using human breast cancer cells and in vivo studies utilizing animal models of human breast cancer. When F. nucleatum was introduced directly into the breast ducts of mice, it triggered significant pathological changes. These included metaplastic and hyperplastic lesions, which are noncancerous cellular abnormalities where cells either proliferate excessively or transform into different cell types. These precancerous changes were accompanied by a surge in inflammation, a critical factor in cancer initiation, and demonstrable DNA damage. Furthermore, the presence of the bacterium significantly increased cell proliferation, a hallmark of cancerous growth.
The impact of F. nucleatum was even more pronounced when it entered the bloodstream and reached pre-existing tumors. In these scenarios, the bacterium dramatically accelerated tumor growth and, alarmingly, promoted the spread of cancer cells to distant organs, specifically the lungs. This metastatic potential is a primary driver of cancer mortality and represents a significant challenge in treatment.
Delving into the molecular underpinnings of these effects, the researchers identified a critical biological process. Exposure to F. nucleatum was found to damage cellular DNA. This damage then activated cellular repair systems. However, the study revealed that these repair systems, particularly a pathway known as nonhomologous end joining (NHEJ), are prone to errors. While NHEJ is essential for rapidly rejoining broken DNA strands, its imperfect nature can introduce mutations. These acquired mutations can then drive further cellular abnormalities and contribute to the development and progression of cancer.
Beyond DNA damage, the bacterium’s presence led to an increase in the levels of a protein called PKcs. This protein was found to be associated with a suite of tumor-promoting behaviors, including enhanced cancer cell motility, increased invasiveness into surrounding tissues, the acquisition of stem-like traits (which confer resistance to treatment and promote recurrence), and a heightened resistance to chemotherapy. This multifaceted impact underscores the bacterium’s ability to fundamentally re-engineer the cellular environment to favor cancer growth and spread.
Genetic Susceptibility: BRCA1 Mutations and Enhanced Risk
The study also shed light on why certain individuals might be more vulnerable to the oncogenic effects of F. nucleatum. Epithelial cells, which form the lining of the breast ducts, and breast cancer cells carrying mutations in the BRCA1 gene were found to be particularly susceptible to the bacterium’s influence.
BRCA1 is a tumor suppressor gene, and mutations in BRCA1 are strongly associated with an increased risk of breast and ovarian cancers. The researchers discovered that BRCA1-mutant cells possess elevated levels of a specific surface sugar, Gal-GalNAc. This sugar molecule acts as a binding site, facilitating the attachment and entry of F. nucleatum into these cells. Consequently, these susceptible cells absorbed significantly more of the bacterium and retained it for extended periods, even across multiple cell generations. This prolonged exposure amplified the DNA damage and cancer-promoting effects initiated by the bacterium.
"Our findings reveal a link between oral microbes and breast cancer risk and progression, particularly in genetically susceptible individuals," Dr. Sharma explained. "Nothing happens in isolation. The results suggest that multiple risk factors come together with F. nucleatum acting as an environmental factor that may cooperate with inherited BRCA1 mutations to promote breast cancer and tumor aggressiveness." This suggests a complex interplay between inherited genetic predispositions and environmental exposures, such as microbial infections, in the development of cancer.
Implications for Public Health and Future Research
The implications of this research are far-reaching, extending from potential diagnostic markers to novel therapeutic interventions and public health recommendations. The identification of a specific bacterium as a contributing factor to breast cancer opens doors for developing diagnostic tests that could assess an individual’s risk based on their oral microbiome. Furthermore, understanding the molecular mechanisms by which F. nucleatum promotes cancer could lead to the development of targeted therapies aimed at neutralizing the bacterium or its harmful effects.
The researchers are cautious but optimistic about the clinical translation of their findings. "More work is needed to understand how these findings translate to patient care," they emphasized. Future studies will focus on a range of critical questions, including the precise prevalence of F. nucleatum in breast tissue of women with and without breast cancer, the efficacy of oral hygiene interventions in potentially mitigating breast cancer risk, and the development of strategies to target the bacterium or its pathways in established cancers.
The current study builds upon a significant body of research over the past decade that has been progressively linking oral health to systemic diseases. For instance, studies published in journals like the Journal of Dental Research and the American Journal of Epidemiology have reported a dose-response relationship between the severity of periodontal disease and the risk of breast cancer. Some studies have also explored the presence of oral bacteria in breast tumor tissue itself, though the causality and mechanisms remained elusive until more recent investigations like the one from Johns Hopkins.
The timeline leading to this discovery can be traced back to the early 2010s when a surge in research began to connect oral pathogens to various systemic conditions, including cardiovascular disease and diabetes. This broader scientific awakening regarding the oral microbiome’s systemic impact created fertile ground for investigating its role in cancer. The Johns Hopkins study, initiated in recent years and culminating in its recent publication, represents a significant leap forward in understanding this complex relationship specifically for breast cancer.
The research team, in addition to Dr. Sharma and Dr. Parida, included Deeptashree Nandi, Deepak Verma, Mingyang Yi, Ashutosh Yendi, Jessica Queen, Kathleen Gabrielson, and Cynthia Sears. Their collective efforts were instrumental in achieving these groundbreaking results.
This extensive investigation was generously supported by funding from the Breast Cancer Research Foundation, Congressionally Directed Medical Research Programs Department of Defense Breast Cancer Research Program grants (BC191572 and BC210668), the John Fetting Fund for Breast Cancer Prevention, and the Bloomberg~Kimmel Institute for Cancer Immunotherapy. This multi-faceted support underscores the recognized importance of this research area and the commitment to unraveling the intricate links between seemingly disparate aspects of human health.
The broader implications for public health are substantial. Encouraging good oral hygiene practices, which are already known to prevent gum disease and tooth loss, may now be viewed as a potential strategy for reducing breast cancer risk. This could involve increased public awareness campaigns and potential integration of oral health screenings into routine cancer prevention check-ups. For individuals with a genetic predisposition, such as those with BRCA1 mutations, maintaining exceptional oral hygiene could become even more critical.
While this research is highly promising, it is crucial to reiterate that correlation does not equal causation, and further clinical trials are necessary to definitively establish the impact of oral health interventions on breast cancer incidence and progression in humans. However, the scientific rigor of the Johns Hopkins study provides a compelling foundation for future research and potential clinical applications. The notion that a common bacterium residing in our mouths could harbor such a profound influence on the development of a major cancer like breast cancer is a stark reminder of the interconnectedness of our body’s systems and the vital importance of maintaining overall health, including that of our oral cavity.