A groundbreaking study published in The Lancet Digital Health has unveiled a remarkable adaptive capacity within the human brain following a stroke, suggesting that even in the face of severe physical disability, undamaged brain regions can exhibit signs of "youthful" structural reorganization. Researchers at the USC Mark and Mary Stevens Neuroimaging and Informatics Institute (Stevens INI), as part of the Enhancing NeuroImaging Genetics through Meta-Analysis (ENIGMA) Stroke Recovery Working Group, have utilized advanced artificial intelligence to analyze brain scans from over 500 stroke survivors worldwide. Their findings indicate that the brain, when confronted with significant injury, can paradoxically rejuvenate certain undamaged areas in an effort to compensate for lost function.
Unveiling the Brain’s Adaptive Strategies
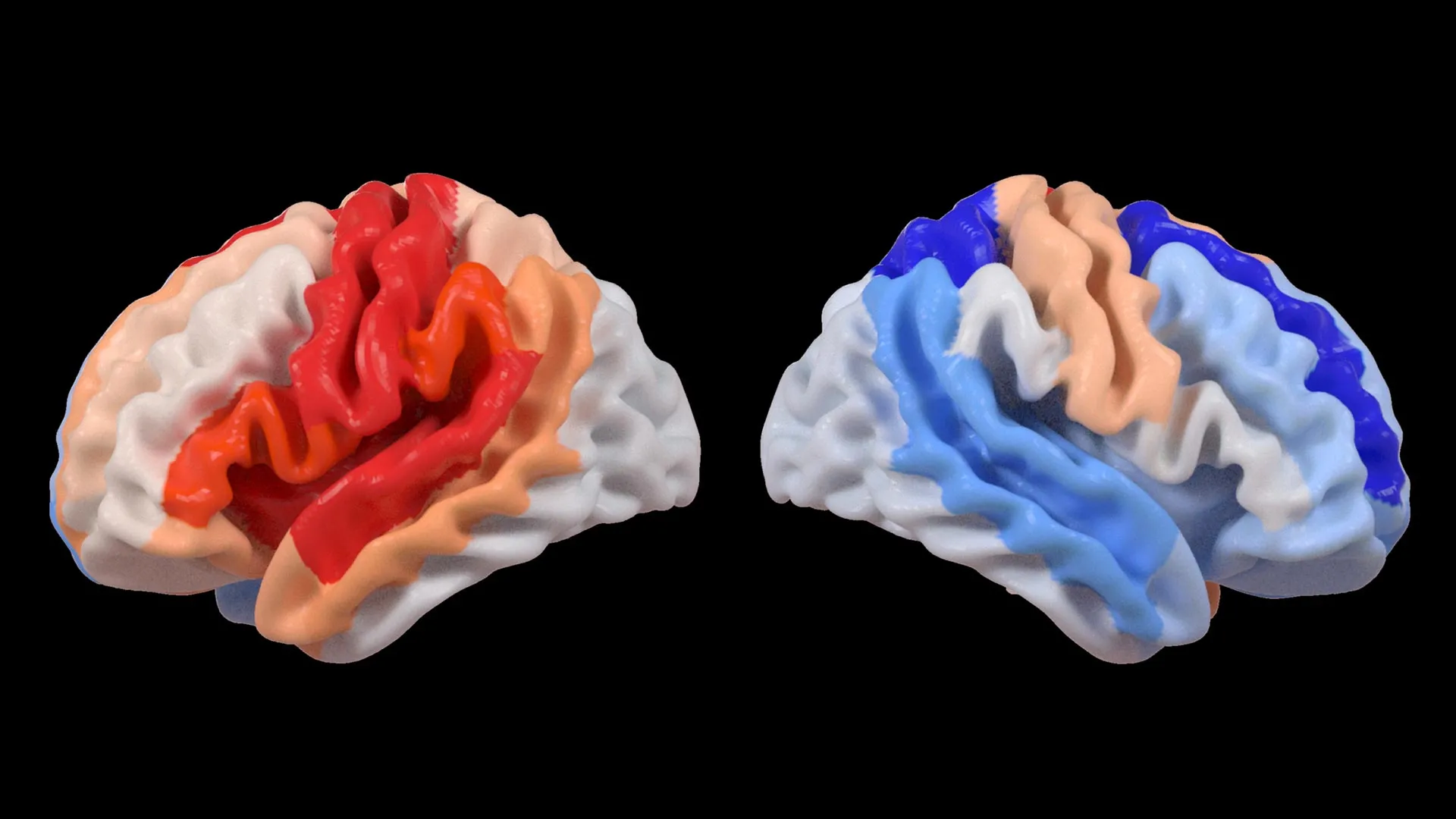
The study’s core revelation centers on the concept of "brain age," a metric derived from sophisticated deep learning models trained on tens of thousands of MRI scans. These models estimate the biological age of specific brain regions. By comparing this estimated age with a participant’s chronological age, researchers can calculate a "brain-predicted age difference" (brain-PAD), which serves as a valuable indicator of brain health and integrity.
A key observation from the ENIGMA data analysis was the differential impact of stroke on brain aging. While the hemisphere directly affected by the stroke often showed accelerated aging, the opposite, undamaged hemisphere displayed a distinct pattern: regions within it appeared younger than expected. This phenomenon was particularly pronounced in individuals who experienced severe physical impairments following their stroke, even after more than six months of intensive rehabilitation.
"We found that larger strokes accelerate aging in the damaged hemisphere but paradoxically make the opposite side of the brain appear younger," stated Hosung Kim, PhD, associate professor of research neurology at the Keck School of Medicine of USC and a co-senior author of the study. "This pattern suggests the brain may be reorganizing itself, essentially rejuvenating undamaged networks to compensate for lost function."
AI as a Window into Neuroplasticity
The sophisticated analysis was made possible by employing a type of artificial intelligence known as a graph convolutional network. This AI system was adept at processing the intricate details within MRI scans, enabling the accurate estimation of biological age for 18 distinct brain regions. The subsequent comparison of these estimated ages with individuals’ actual ages allowed for the calculation of brain-PAD scores.
When these brain-PAD scores were correlated with motor function assessments, a compelling pattern emerged. Stroke survivors exhibiting significant motor deficits, even those who had undergone extensive rehabilitation, consistently showed a younger-than-expected brain age in regions of the hemisphere contralateral to the stroke lesion. This effect was especially prominent within the frontoparietal network, a critical brain system involved in planning movements, directing attention, and coordinating actions.
"These findings suggest that when stroke damage leads to greater movement loss, undamaged regions on the opposite side of the brain may adapt to help compensate," Kim elaborated. "We saw this in the contralesional frontoparietal network, which showed a more ‘youthful’ pattern and is known to support motor planning, attention, and coordination. Rather than indicating full recovery of movement, this pattern may reflect the brain’s attempt to adjust when the damaged motor system can no longer function normally. This gives us a new way to see neuroplasticity that traditional imaging could not capture."
This discovery offers a novel perspective on neuroplasticity, the brain’s remarkable ability to reorganize itself by forming new neural connections throughout life. Traditionally, neuroplasticity has been observed through changes in brain activity or the growth of new neurons. However, this study highlights a more subtle, structural form of adaptation reflected in the biological aging patterns of brain tissue. The "youthful" appearance in contralateral regions may signify heightened metabolic activity, increased synaptic density, or a more efficient cellular state, all indicative of the brain actively working to reroute and compensate for compromised pathways.
The Power of Global Collaboration: The ENIGMA Initiative
The success of this research is deeply rooted in the ENIGMA initiative, a vast international consortium that aggregates neuroimaging data from across the globe. ENIGMA’s mission is to enhance our understanding of the brain in health and disease by creating standardized datasets and fostering collaborative research. The Stroke Recovery Working Group, a key component of ENIGMA, specifically focuses on unraveling the complexities of stroke recovery.
By pooling data from numerous research centers spanning more than 50 countries, the ENIGMA Stroke Recovery Working Group has amassed the largest stroke neuroimaging dataset of its kind. This immense collection of MRI scans and associated clinical information, meticulously standardized, provides an unprecedented opportunity to detect subtle patterns that might remain hidden in smaller, single-center studies.
"By pooling data from hundreds of stroke survivors worldwide and applying cutting-edge AI, we can detect subtle patterns of brain reorganization that would be invisible in smaller studies," commented Arthur W. Toga, PhD, director of the Stevens INI and Provost Professor at USC. "These findings of regionally differential brain aging in chronic stroke could eventually guide personalized rehabilitation strategies."
The timeline for data collection and analysis within the ENIGMA framework is extensive, often spanning years. Individual studies contributing to ENIGMA may have initiated data collection as far back as the early 2000s, with ongoing efforts to include newer cohorts. The standardization process itself is a significant undertaking, involving the harmonization of imaging protocols, data quality control, and the development of common analytical pipelines. This rigorous approach ensures that the insights derived from the combined dataset are robust and generalizable.
Implications for Stroke Rehabilitation and Future Research
The findings from this study carry profound implications for the future of stroke rehabilitation. Understanding the specific ways the brain reorganizes itself after injury can pave the way for more targeted and personalized treatment strategies. If certain undamaged brain regions are actively working to compensate for lost motor function, therapeutic interventions could be designed to further support and enhance these adaptive processes.
For instance, rehabilitation programs might be tailored to optimize the function of the contralesional frontoparietal network in patients with severe motor impairments. This could involve specific types of exercises, cognitive training, or even neuromodulation techniques aimed at bolstering the neural pathways within this network. The "younger" brain age observed in these regions suggests a heightened potential for plasticity, making them prime targets for therapeutic enhancement.
The researchers are not resting on their laurels. Their future plans involve longitudinal studies, tracking stroke survivors from the acute stages of recovery through to long-term outcomes. By observing how brain aging patterns and structural changes evolve over time, clinicians could gain invaluable insights into an individual’s unique recovery trajectory. This dynamic understanding is crucial for adapting rehabilitation plans as the patient progresses and their brain continues to adapt.
"The goal is to move beyond one-size-fits-all approaches and develop therapies that are precisely matched to each patient’s individual neurobiological profile," said Dr. Kim. "This study represents a significant step in that direction, offering a new biomarker for assessing brain adaptation and recovery potential."
The study’s reliance on a global dataset also underscores the importance of international collaboration in advancing medical science. Stroke is a global health challenge, affecting millions of people annually. By sharing data and expertise across borders, researchers can accelerate the pace of discovery and bring innovative treatments to patients more quickly. The NIH grant R01 NS115845, which funded this research, highlights the significant investment required for such large-scale, international endeavors. The participation of institutions like the University of British Columbia, Monash University, Emory University, and the University of Oslo further emphasizes the collaborative spirit driving this research.
The potential for this research to impact patient care is substantial. Currently, prognoses for motor recovery after stroke can be challenging to predict with certainty, especially in cases of severe impairment. The brain age metrics derived from this study could potentially serve as a predictive tool, helping clinicians set realistic expectations and tailor rehabilitation intensity and duration. Furthermore, it may offer a way to identify individuals who could benefit most from emerging neurorehabilitative technologies, such as brain-computer interfaces or advanced forms of physical therapy.
The study, titled "Deep learning prediction of MRI-based regional brain age reveals contralesional neuroplasticity associated with severe motor impairment in chronic stroke: A worldwide ENIGMA study," has been published in The Lancet Digital Health, a leading journal in the field of digital health and artificial intelligence in medicine. This publication ensures that the findings are accessible to the global scientific and medical community, fostering further research and clinical application.
In conclusion, this pioneering work from the USC Stevens INI and the ENIGMA consortium has illuminated a fascinating aspect of the post-stroke brain: its inherent ability to adapt and potentially "rejuvenate" undamaged areas to compensate for injury. By harnessing the power of artificial intelligence and large-scale international collaboration, researchers have uncovered a novel pathway to understand neuroplasticity, offering hope for more personalized and effective stroke recovery strategies in the future. The implications of this research extend beyond mere scientific curiosity, promising to directly benefit stroke survivors by optimizing their rehabilitation journeys and ultimately improving their quality of life. The ongoing work to track these brain changes over time will be critical in translating these profound discoveries into tangible clinical advancements.